How does it work?

Each time you brush your teeth, teeth absorb some of the fluoride. This fluoride makes the enamel prevent the demineralization, which occurs when acids from bacteria leach calcium and phosphates from your teeth, weakening the enamel, thus setting a stage for cavities (Severson). By applying fluoride and strengthening the enamel, bacteria acids aren’t able to leach minerals form your teeth, helping prevent decay and cavities. The three main jobs of toothpaste are to prevent cavities, remove plaque and stop gingivitis.
The most essentials ingredient used is fluoride –surfactants work well with fluoride to form foam that clean the tooth surface and wash away the plaque. Also, tetrasodium pyrophosphate (TSSP) is an ingredient used in toothpaste that helps remove magnesium and calcium from our saliva because they form tartar or plaque on our teeth. In order to reduce the bitterness of TSSP, they often add sodium saccharin as sweetener.
The surface of the teeth is enamel and is 95% made from hydroxyapatite which is: Ca10(PO4)6(OH)2
Although hydroxyapatite is strong and hard, it can be attacked by acid and causes chemical reaction:
Ca10(PO4)6(OH)2 + 8H+ ↔ 10Ca2+ + 6(HPO4)2- + 2H2O
This equation means that the more Acid (H+ion) is added to the enamel, and the more hydroxyapatite is dissolved into saliva. It is called “decalcification”. Eventually, the enamel will start to decay, and plaque is formed. This is how the teeth will be without toothpaste.
The most essentials ingredient used is fluoride –surfactants work well with fluoride to form foam that clean the tooth surface and wash away the plaque. Also, tetrasodium pyrophosphate (TSSP) is an ingredient used in toothpaste that helps remove magnesium and calcium from our saliva because they form tartar or plaque on our teeth. In order to reduce the bitterness of TSSP, they often add sodium saccharin as sweetener.
The surface of the teeth is enamel and is 95% made from hydroxyapatite which is: Ca10(PO4)6(OH)2
Although hydroxyapatite is strong and hard, it can be attacked by acid and causes chemical reaction:
Ca10(PO4)6(OH)2 + 8H+ ↔ 10Ca2+ + 6(HPO4)2- + 2H2O
This equation means that the more Acid (H+ion) is added to the enamel, and the more hydroxyapatite is dissolved into saliva. It is called “decalcification”. Eventually, the enamel will start to decay, and plaque is formed. This is how the teeth will be without toothpaste.
How can cavity be prevented?
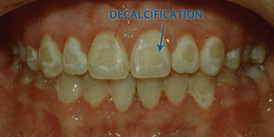
Most food we eat is made from different types of acid such as malic acid (apple), tannic acid(tea) and tartaric acid (carrot). The toothpaste act as base to neutralize the acid in our mouth. Since the acids attack the enamel and diffuse into calcium ion and HPO4 2- ions from the equation, so when we brush our teeth with toothpaste, fluoride ions are delivered to the enamel and replaces the OH ions in hydroxyapatite, to form fluoroapatite (Ca10(PO4)6F2), which would eventually give protection to the teeth (Kelvin and Yon). The equation of this reaction is:
Ca10(PO4)6(OH)2 + 8H would turn into 10Ca2+ + 6HPO42 + 2H2O
(Kevin and Yon).
Ca10(PO4)6(OH)2 + 8H would turn into 10Ca2+ + 6HPO42 + 2H2O
(Kevin and Yon).
